Equipment
In Hot Water: Do Active Heating Systems Increase The Risk of DCI?
Active thermal heating systems will keep you warm in cold water and improve diver safety, but can they also increase your risk of getting bent? Our science geek Reilly Fogarty reviews the current conflicting research, teases out the factors involved, and identifies a ‘Best Practices’ strategy for those intrepid souls preparing to make the jump into very cold water.

by Reilly Fogarty
Header image: A Navy diver undergoing a thermal experiment at the US Navy Experimental Diving Unit (NEDU) in Panama City, Fl. Photo by Stefan Frink
Heating solutions for divers have come a long way in the past two decades. Not long ago a diver’s only option for active heating underwater was a hot water suit, complete with the logistical limitations of surface or habitat supplied water. Equipment and tenders made the suits difficult to put to use and divers were limited to exploring the seafloor only as far as their hot water hose could reach. These challenges made active heating accessible only to a small subset of the commercial diving industry, and nearly impossible for recreational divers to use except for some notable but extraordinarily rare exceptions.

Limited by thermal exposure on long dives in warm water and short excursions in wintry waters alike, recreational divers began using hot-packs, hot water bottles, and any number of similar solutions to heat their drysuits with typically underwhelming results. The evolution of electrical heating systems however, brought active heating within reach for a huge number of new divers. The first iterations were fickle and expensive, and many home-brew solutions involving heated motorcycle vests and DIY-battery packs were concocted, but the recreational industry adopted the technology in relatively short order. Now divers have a wealth of options for heated undergarments, from minimalist and self-contained systems that wear like a t-shirt and can be used in a wetsuit to full-body undergarments with gloves and booties powered by external battery packs.

Recreational divers have adopted these heated undergarments rather quickly, but the total market share is not yet widespread enough to allow much in the way of statistical analysis. The technology has made diving a winter sport for many, but the proliferation without adequate research has brought some serious concerns about decompression stress to the forefront. While these new tools make it possible to comfortably endure harsher climates and longer exposures, if they are used incorrectly, they can dramatically increase decompression stress. Here’s what you should know as you decide whether to heat your next dive.
Heating’s Double Edged Sword
The crux of the concern surrounding heated undergarment use lies in the effect of temperature on decompression sickness risk. A number of aptly named studies have laid the statistical groundwork for what most of us already believe to be true—temperature has a significant effect on our ability to absorb and eliminate inert gases.
Cold immersion gives us a number of things to contend with. Vasoconstriction, the narrowing of blood vessels, helps shunt blood to the core to maintain core temperature but leaves hands and feet cold and quick to numb. Slow perfusion in these tissues can slow both the uptake and removal of inert gases, the latter of which increases decompression stress. The body will also attempt to eliminate fluid via urination, promoting dehydration, and in some cases breathing rate can be increased via cold water shock or increased metabolic drive to keep the body warm. These factors are familiar to divers, but they all contribute to decompression stress.
Active heating systems—used properly—can address these factors. Keeping a diver warm can minimize vasoconstriction and improve perfusion, improving inert gas elimination in extremities. Warm divers will shunt less fluid to their core, produce less urine and face fewer concerns from cold-induced physiological reactions. It’s the potential to increase total inert gas load via warming throughout a dive, or allowing efficient gas loading and then hampering decompression via the failure or removal of heat on the ascent portion of a dive that can put a diver at serious risk. Not only are these devices prone to failure just by nature of being electrical in an underwater environment, but even properly functioning, their inappropriate application can leave a diver with significantly more decompression stress than they would have faced on a dive without their heating solution of choice.

It’s the severity of these risks in the real world coupled with the conflicting data that makes this a tough topic to tackle for recreational divers. Heated undergarments make an enormous difference on long technical dives, they have the potential to make a dive not only more comfortable but safer, but they can also put divers in needless risk.
Conflicting Research
Even the best data on thermal status fails to give us more than correlations with DCS symptoms, which makes estimating risk nearly impossible. There is, however, a respectable body of research that indicates divers using hot water suits may experience DCS at a higher rate than their counterparts. One study from 1951 on hot water suit use among surface decompression dives indicated that each 10°C increase in water temperature increased the odds ratio of DCS by 1.96 and that this effect was most pronounced on shorter dives in the study. A later review of that study however, indicated that the probability of some of the DCS symptoms, specifically the Type 2 symptoms could have been “better explained by the dive profile than by the temperature” (Leffler, 2001). Another work by the same author indicates a significant increase in DCS risk among divers who are warm at depth, specifically pointing to vasodilation induced promotion of on-gassing efficiency. This correlation between hot water suit use at depth and DCS risk was also found among divers working on the TWA Flight 800 recovery in 1997.

It’s worth noting that hot water suits and electrical heated undergarments may not be entirely identical systems. Hot water suits have a much greater heating potential and have been shown to cause some fluid loss in divers, primarily from sweat. In real-world applications however, both can be used in a similar enough fashion that many of the lessons learned from research into hot water suit use can be carried over to more modern systems.
The real conflict in data and theory comes in both the application of the heating systems, and the balance of heat needed to maintain dexterity and complete a mission, and decompression risk. Even working from a foundation of data that suggests the following:
- Being warm during a dive increases post-dive bubble scores
- Hot water suits are associated with higher DCS risk
- Post-dive cooling could prolong the period of elevated risk for Type 1 DCS
There is still some room for the safe application of active heating to improve both safety and comfort. A 2007 NEDU study showed a significant decrease in DCS incidence in a group of divers performing a 150fsw/60 minute dive on U.S. Navy Standard Air tables that were kept cold during compression phases (descent and bottom time) and warmed during decompression, compared to a group from a prior study that was kept cold throughout their dive, despite the “cold” group decompressing for nearly 2.5 times as long.
This study was able to create a dataset that included more than 400 dives to a depth of 120 feet of seawater, a standard decompression profile and varying thermal exposures, providing a profile that can be reasonably extrapolated to recreational profiles. The principal of this study was the comparison of DCS incidence odds ratios between these thermal exposures, resulting in a 23.8% DCS odds ratio for a 10C increase in temperature during compression, and decreased DCS occurrence and VGE scores (although postdive VGE scores were only weakly associated with DCS occurrence).
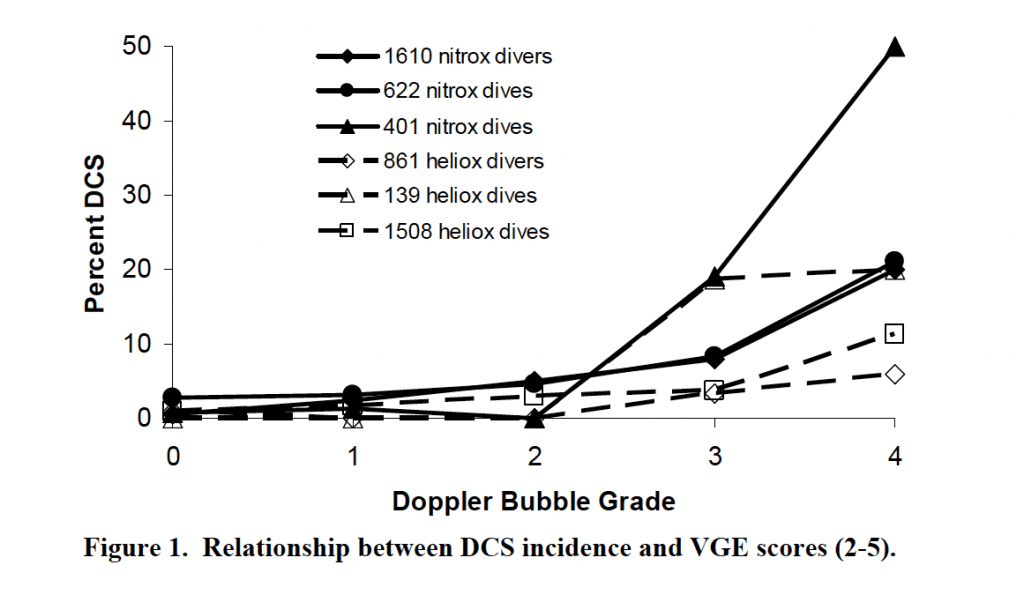
A group of physicians and researchers did take issue with some of the results, hoping to temper recreational divers from extrapolating data directly, but their editorial was not without rebuttal from their colleagues. The study’s authors eventually waded into the academic dispute with their own response clarifying that while the thermal stresses experienced by recreational divers are likely less than found in his experiment, the responses would likely be similar but in lesser magnitude. Because of this, they contend that it would be unwise to ignore the trends they found, and the data could have a profound effect on the larger diving community, remarking that, “We wish to clarify that our study does have implications for recreational and technical divers, implications that should not be ignored.”
Much in the way that the original U.S. Navy Dive Tables were adapted for the recreational market, so too can this data provide valuable lessons to divers who do not necessarily resemble the hyper-fit Navy Dive standard.
Best Practices
With the limited data we have and the considerable academic dispute over the cumulative effects of various heating applications, it seems that the best course of action is to draw from a combination of the NEDU study, community engrained platitudes about thermal status, and a healthy dose of theoretical modeling. Pollock, Clark et. al, and the NEDU all agree at some level that active heating can be applied to improve divers safety. In this application, the NEDU study would seem to indicate that the most appropriate application would be to keep divers cool during the compression phases (during descent and the working portion of the dive), and gently warm them during the ascent to aid in decompression. In situations where the working portion of the dive requires heating at a “minimal level” can likely be safely applied, but excessive heating on ascent should be avoided to prevent dehydration or decompression that is too aggressive.

Logistically, this addresses the worst-case scenario, a diver intending to run active heating throughout a dive who experiences a failure on ascent, leading to a warm compression and cold decompression phase. It also makes it possible to use heating to some extent in the exploration of harsh environments and maintain comfort and dexterity not possible with passive thermal protection. It does not, however, address the fact that decompression models do not account for thermal status, let alone change in thermal status during a dive. Additional conservatism must be applied by the diver, because the addition of an active heating system provides one more variable in amongst the milieu of uncertainty in decompression risk.
Using heated undergarments in this “cold/warm” fashion seems to be the takeaway for Pollock, the NEDU, and many of their colleagues. The NEDU study goes so far as to say that their group following the “cold/warm” pattern experienced a benefit similar to halving their bottom time, compared to the group that was kept cold throughout the dive and decompressed for 2.5 times as long. The potential for enormous benefit is there, but applied incorrectly it seems likely that the opposite is also true. “Dramatic results demand serious attention” is how Pollock put it, and it’s worth keeping that in mind as you weigh your options, and your wallet, this spring.
References:
1. Effect of ambient temperature on the risk of decompression sickness in surface decompression divers
2. Effect of ambient temperature on the risk of decompression sickness in surface decompression divers
3. Recompression treatments during the recovery of TWA Flight 800
4. Time and temperature effects on body fluid loss during dives with the open hot-water suit
5. Re: Don’t dive cold when you don’t have to (Pollock)
6. The Influence of Thermal Exposure on Diver Susceptibility to Decompression Sickness
7. Don’t Dive Cold When You Don’t Have To (TDI)
8. On diver thermal status and susceptibility to decompression sickness (letter)
9. Thermal stress and diver protection.
Dive Deeper:
Alert Diver: Deep in the Science of Diving: The Navy Experimental Diving Unit by Michael Menduno

Reilly Fogarty is a team leader for risk mitigation initiatives at Divers Alert Network (DAN). When not working on safety programs for DAN, he can be found running technical charters and teaching rebreather diving in Gloucester, MA. Reilly is a USCG licensed captain whose professional background includes surgical and wilderness emergency medicine as well as dive shop management.